Pre-treatment prior to membranes using AFM®
Introduction
The pretreatment of water prior to membranes is a critical first stage that can make a significant difference to the economics, sustainability and ease of operation of an RO water treatment system. RO membranes for desalination or TDS reduction will always be subject to fouling from biological contamination, organic and inorganic chemical precipitation. Pretreatment usually involves sand filters or Ultra Filtration (UF), followed by 5-micron and 1-micron cartridge filters and activated carbon or UVc irradiation. There are operational costs associated with the pretreatment process and major implications with regards to the security and protection of the membranes. The membranes must be allowed to perform their proper function without excessive demand for maintenance and cleaning chemicals. AFM® will reduce the risks, reduce the costs and simplify the process.

Disadvantages of current pre-treatment technologies
Ultra filtration (UF) down to 0.03um
UF has better mechanical filtration performance than sand / cartridge filter combination, but UF will not remove dissolved organics or chemicals from solution. UF is purely a mechanical filtration process, dissolved components, or particles smaller than 0.03 microns will pass trough the membranes. The dissolved organics lead to biofouling of the membranes. The inorganic components such as free silica or phosphate will form a precipitate and scale up the membranes.
Sand filtration followed by cartridge filters
Sand is effective at removing particulates and dissolved biological nutrients, but the filter will generate bacterial cell biomass, which will foul the membranes. Sand filters also suffer from biodynamic instability leading to transient wormhole channelling and passage of unfiltered water which blocks the cartridge filters. This process takes about 6 months before it starts to impact on system performance. Coagulants and flocculants maybe used prior to sand filters to remove phosphate, but sand has free silica and this will cause a blockage of the membranes, especially if there is aluminium in the water or if aluminium is used as a flocculent.
AFM® filtration as a pre-treatment prior to RO
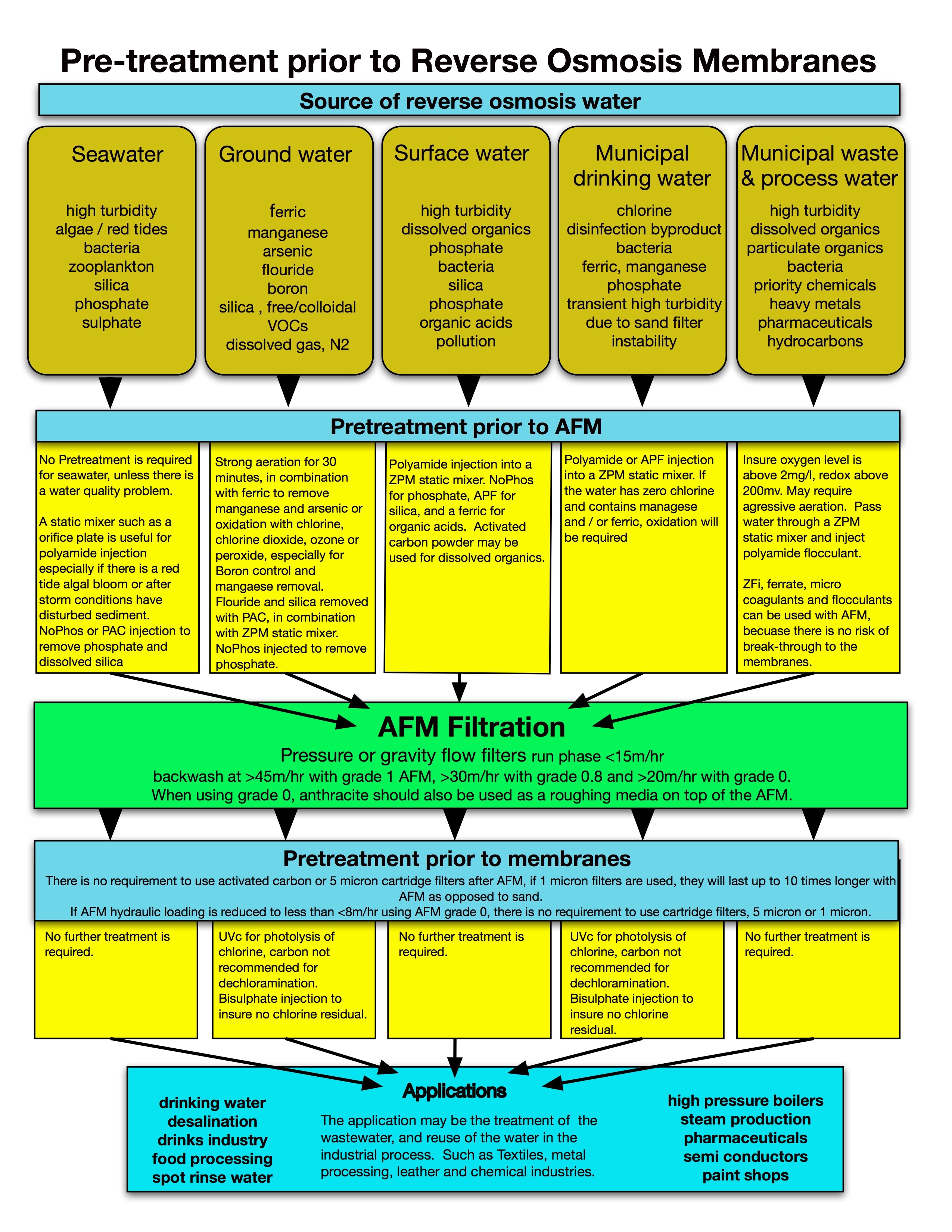
AFM® is an activated mesoporous aluminosilcate with glass as a structural substrate and a direct replacement for sand with similar operational criteria. AFM® has a surface area that is up to 300 times greater than sand. The very large surface area has a negative –ve charge for the adsorption of positive +ve particles that are too small to be mechanically removed from the water. When AFM® is combined with pre-coagulation and flocculation, mechanical filtration performance is improved by up to 100 times to a nominal filtration down to 0.1 microns. In addition to removing solids, the coagulation reactions will remove dissolved organics such as proteins, lipids, amino acids and inorganic components including phosphate and free silica. AFM® is also excellent at removing hydrocarbons. AFM® performance has been independently verified by IFTS (Institute of Filtration and Technical Separation) in France in 2014. See details on pretreatment prior to AFM® and pretreatment prior to RO in the chart to the right.
Pre-treatment prior to AFM® filtration
Depending on the raw water source, a pretreatment prior to the AFM® filtration is necessary. For example: seawater or high TDS water abstracted from the ground via a borehole (tube-well) water, often has the following conditions:
- no oxygen and low redox potential
- high zeta potential and colloidal suspension of silica
- elevated partial pressure of nitrogen gas
- dissolved sulphide, methane and VOC's (volatile organics carbon)
- iron, manganese & arsenic
- heavy metals such as chromium, lead, mercury
- phosphate
- colloidal silica
- Fluoride
- Ammonium, in combination with phosphate and magnesium if forms struvite
If the water has dissolved oxygen content below 5mg/l, or a redox potential under 200mv, the very first step should be aeration to gas strip the water and restore the chemical equilibrium.
Gas stripping will blow off volatile components and stabilize the alkalinity, this is essential for efficient filtration and to allow the zeta potential of the water to drop. The aeration phase should be from 10 to 30 minutes. During aeration, the oxidation potential will increase and most heavy metals will precipitate out of solution. With regards to iron, it will change from Fe2+ to Fe3+, arsenic changes from AS3+ to AS5+.
During the oxidation stage with Fe2+ to Fe3+, the small ferric particles will be suspended by the aeration will grow and react to co-precipitate other heavy metals from solution. The longer the aeration phases the larger the ferric particles making them easier to be remove by AFM®. In some cases we recommend the addition of ferric to the water in order to assist with co-precipitation reactions.
AFM® key performance benefits
- Particle removal of 97% down to 5 microns with Grade 1 and 99.7% down to 1 microns with Grade 0 independently verified
- When combined with coagulation and flocculation, nominal filtration performance is better than 0.1 microns
- AFM® will deliver a 50% to 75% lower SDI than a sand filter
- Unlike sand there is no free silica in AFM®, so less fouling of membranes
- Performance comparable to UF
- Does not biofoul or channel
- AFM® directly adsorbs organics like activated carbon
- Chlorination is not required, so no TCA, THM or hydrobromous acid production
- Regenerated by back-washing with water
- Perfect for ground water filtration and removal of heavy metals
Webdesign by Bönzli + Feuz IT
